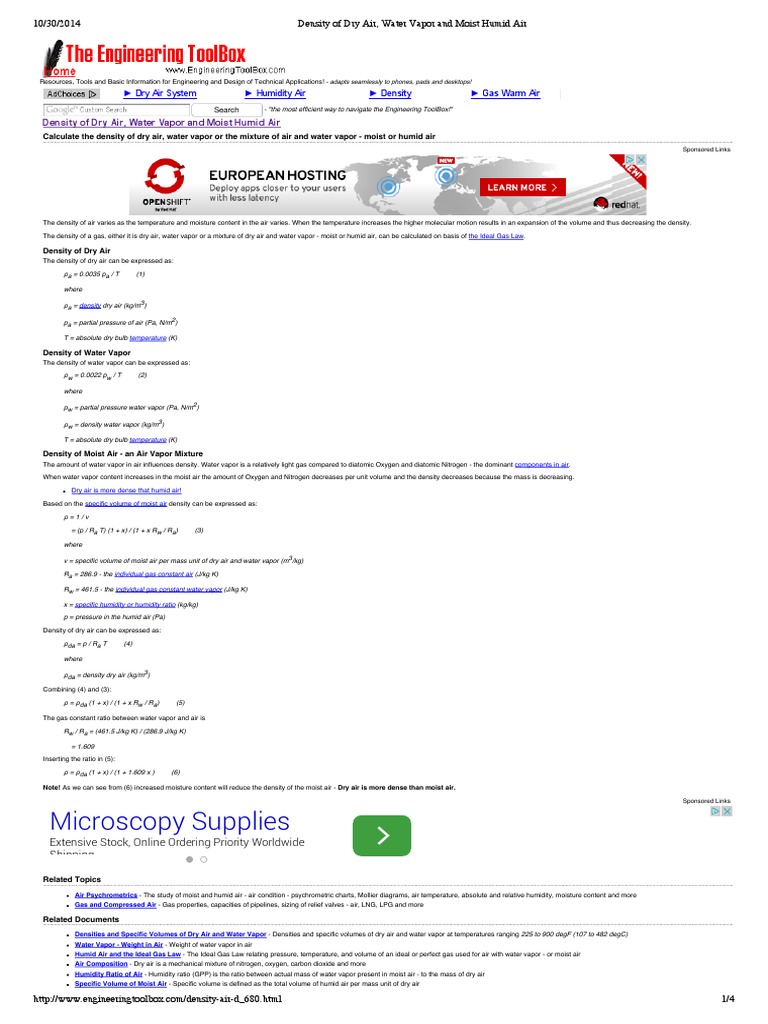
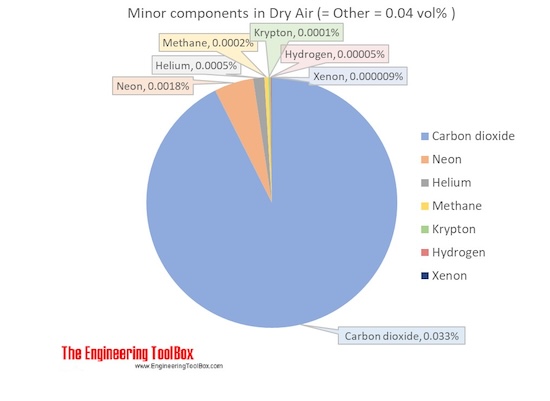
AOS 101 February 12 or 14 Ideal Gas Law. P = pressure (in Pascals) ρ = density (in kg/m 3 ) = mass / volume R = gas constant (dry air: R = 287 J/kg K) - ppt download

thermodynamics - why sometimes we can find p=ρgRT instead of p=ρRT for ideal gas? - Physics Stack Exchange
![What is the density of wet air with 75% relative humidity at 1 atm and 300 K? [Given: vapour pressure of H2O is 30 torr and average molar mass of air is 29 g mol^-1 ] What is the density of wet air with 75% relative humidity at 1 atm and 300 K? [Given: vapour pressure of H2O is 30 torr and average molar mass of air is 29 g mol^-1 ]](https://dwes9vv9u0550.cloudfront.net/images/6059330/4edb35d6-6335-4801-a33e-56f47d3e5cd3.jpg)
What is the density of wet air with 75% relative humidity at 1 atm and 300 K? [Given: vapour pressure of H2O is 30 torr and average molar mass of air is 29 g mol^-1 ]
Introduction to Atmosphere and Space Science Prof. M. V. Sunil Krishna Department of Physics Indian Institute of Technology, Roo