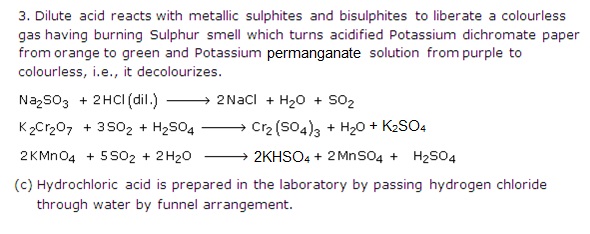
Sulfuric acid (H2SO4) is added to an unknown precipitate to induce a chemical reaction revealing the indentity of the metal in question. Orange color indicates iron Stock Photo - Alamy

Fe2O3+H2SO4=Fe2(SO4)3+H2O Balanced Equation|| Balanced equation for Iron iii oxide and Sulfuric acid - YouTube

Iron in sulphuric acid. Bubbles of hydrogen forming on iron powder as they react with weak sulphuric acid in a test tube. This reaction forms a colour Stock Photo - Alamy

How to Balance Fe + H2SO4 = FeSO4 + Fe2(SO4)3 + H2O + SO2 (Iron + Concentrated Sulfuric acid) - YouTube

inorganic chemistry - How to get back my beautiful crystals made from iron and sulphuric acid - Chemistry Stack Exchange

Question Video: Identifying the Balanced Symbolic Equation for the Reaction of Iron Metal with Concentrated Sulfuric Acid | Nagwa

Effect of sulfuric acid concentration on iron dissolution and final pH.... | Download Scientific Diagram