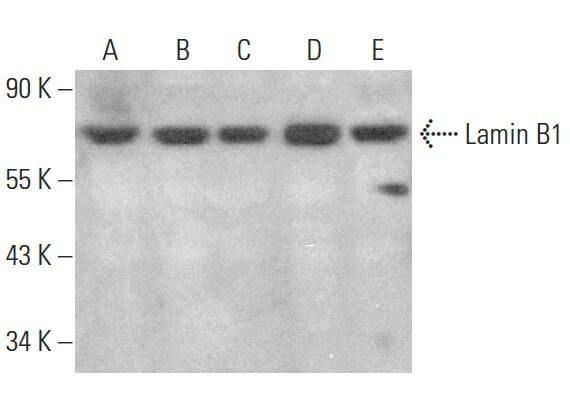
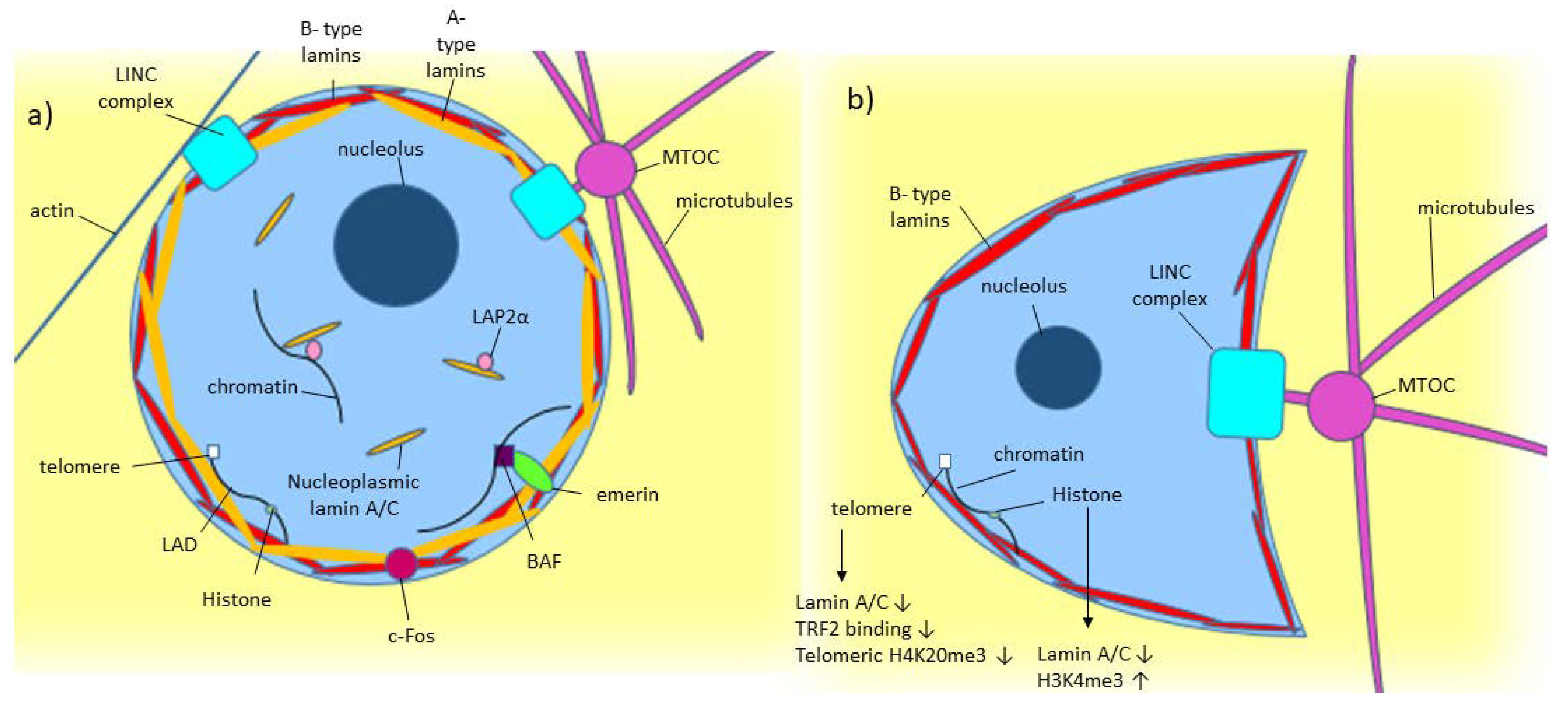
Tyrosine phosphorylation of lamin A by Src promotes disassembly of nuclear lamina in interphase | Life Science Alliance

IJMS | Free Full-Text | Quantification of the Lamin A/C Transcript Variants in Cancer Cell Lines by Targeted Absolute Quantitative Proteomics and Correlation with mRNA Expression
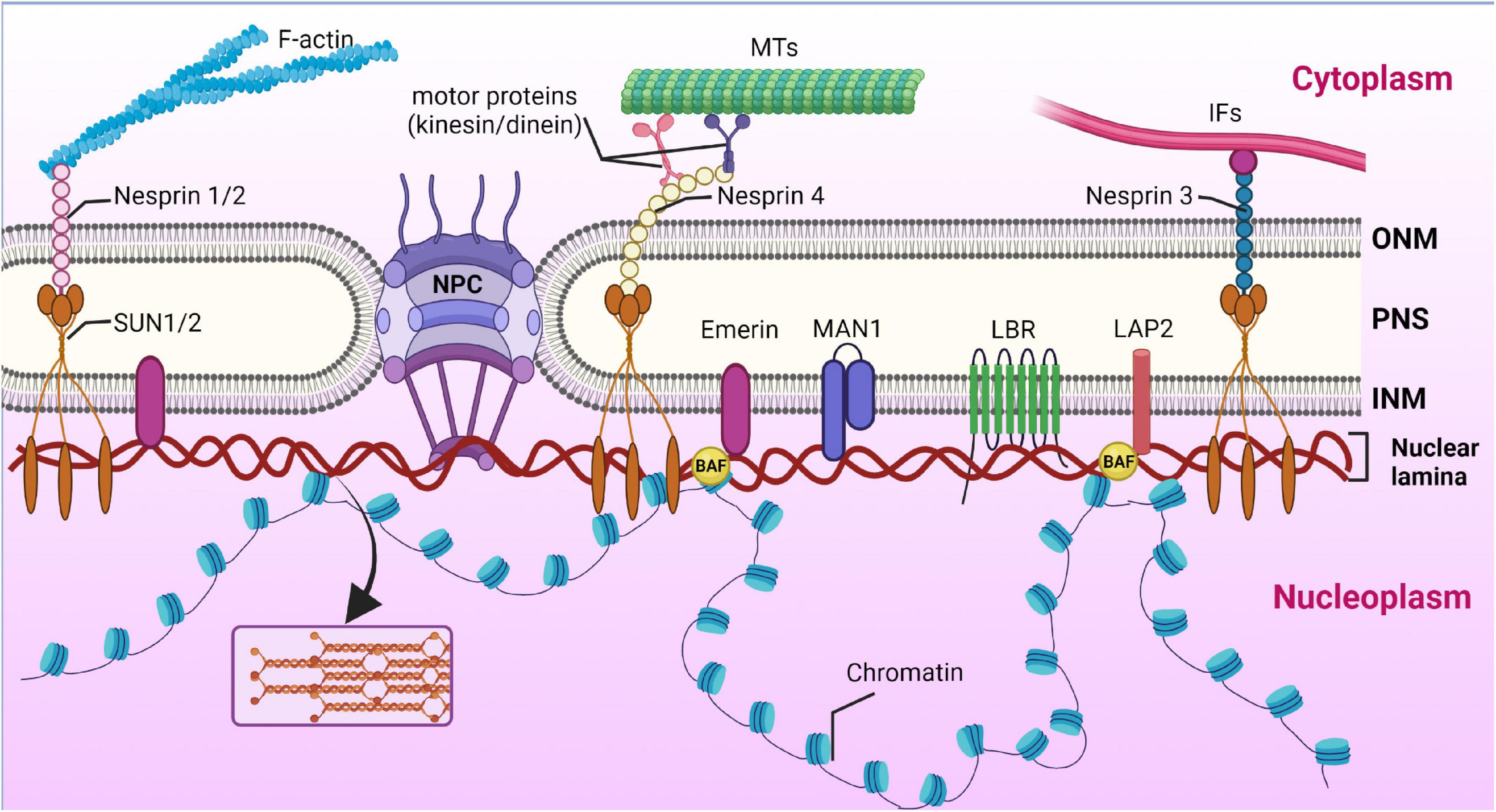
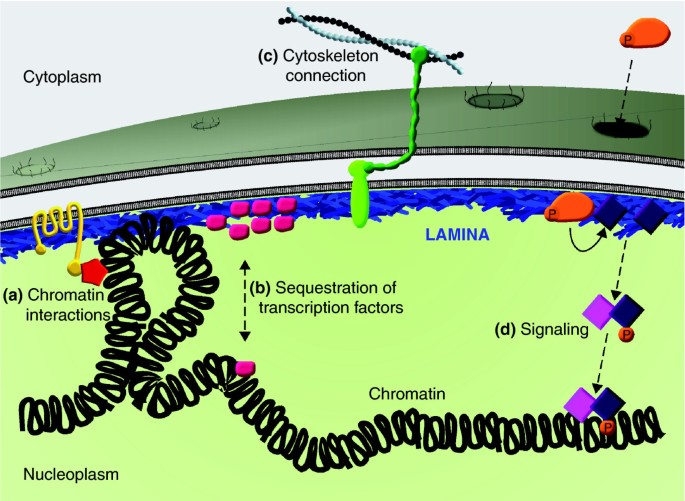
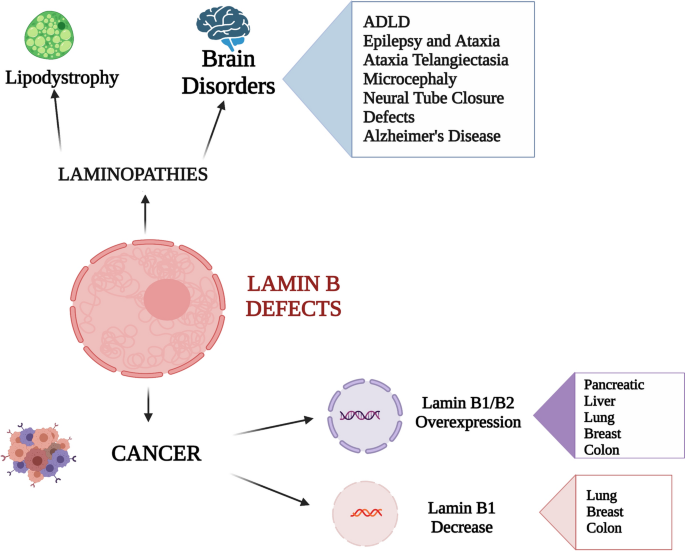
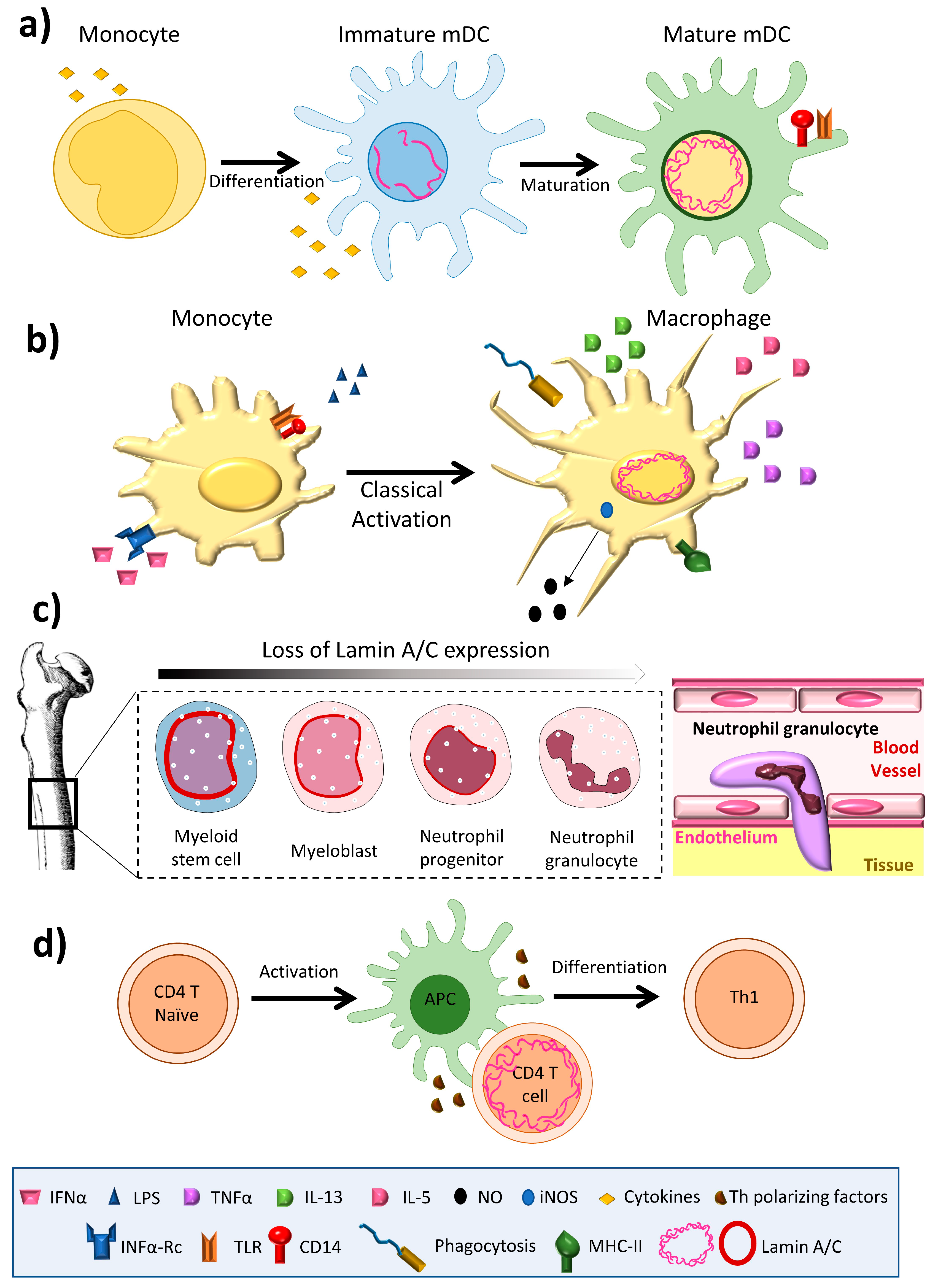
Frontiers | Diversity of Nuclear Lamin A/C Action as a Key to Tissue-Specific Regulation of Cellular Identity in Health and Disease
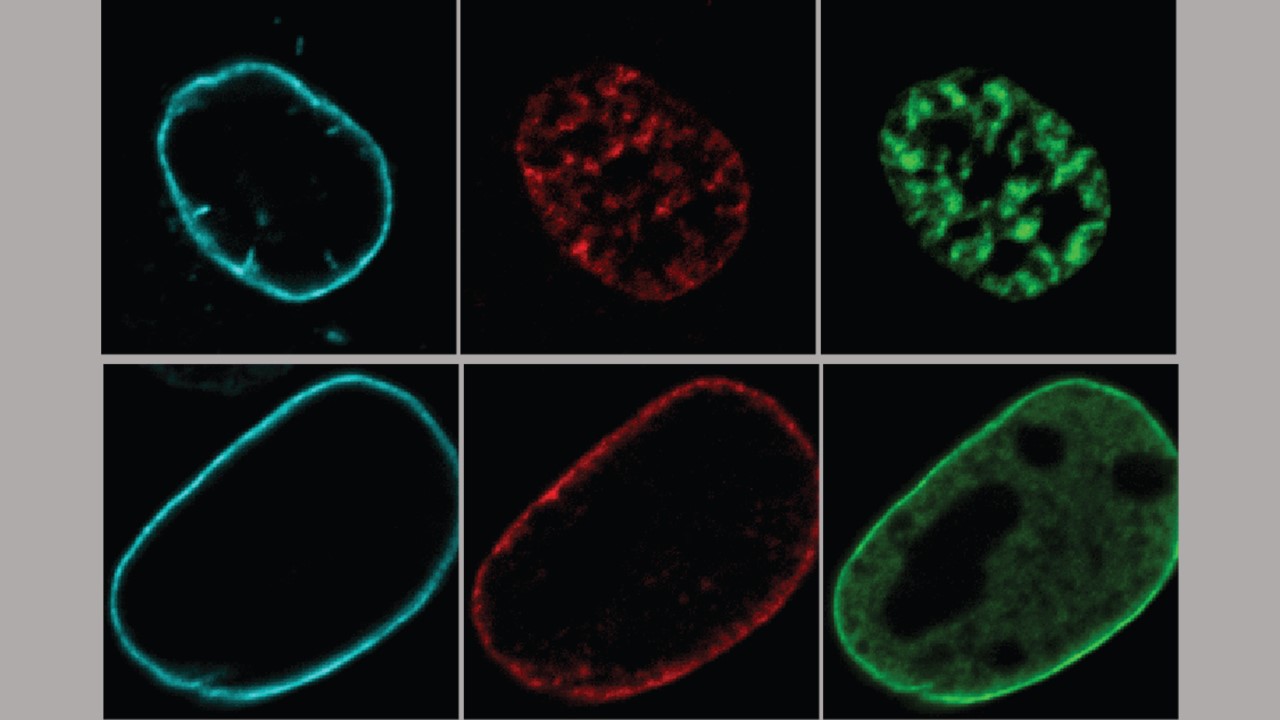
Mouse Cell Studies Show That Correcting DNA Disorganization Could Aid Diagnosis and Treatment of Rare Inherited Diseases

Phosphorylated Lamin A/C in the Nuclear Interior Binds Active Enhancers Associated with Abnormal Transcription in Progeria - ScienceDirect

Phosphorylated Lamin A/C in the Nuclear Interior Binds Active Enhancers Associated with Abnormal Transcription in Progeria - ScienceDirect

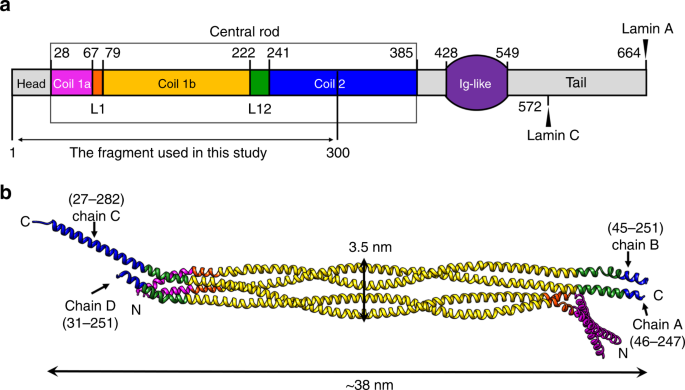
Comparison of lamin A, lamin C, and LA ⌬ 50 structures. ( A ) The 12... | Download Scientific Diagram
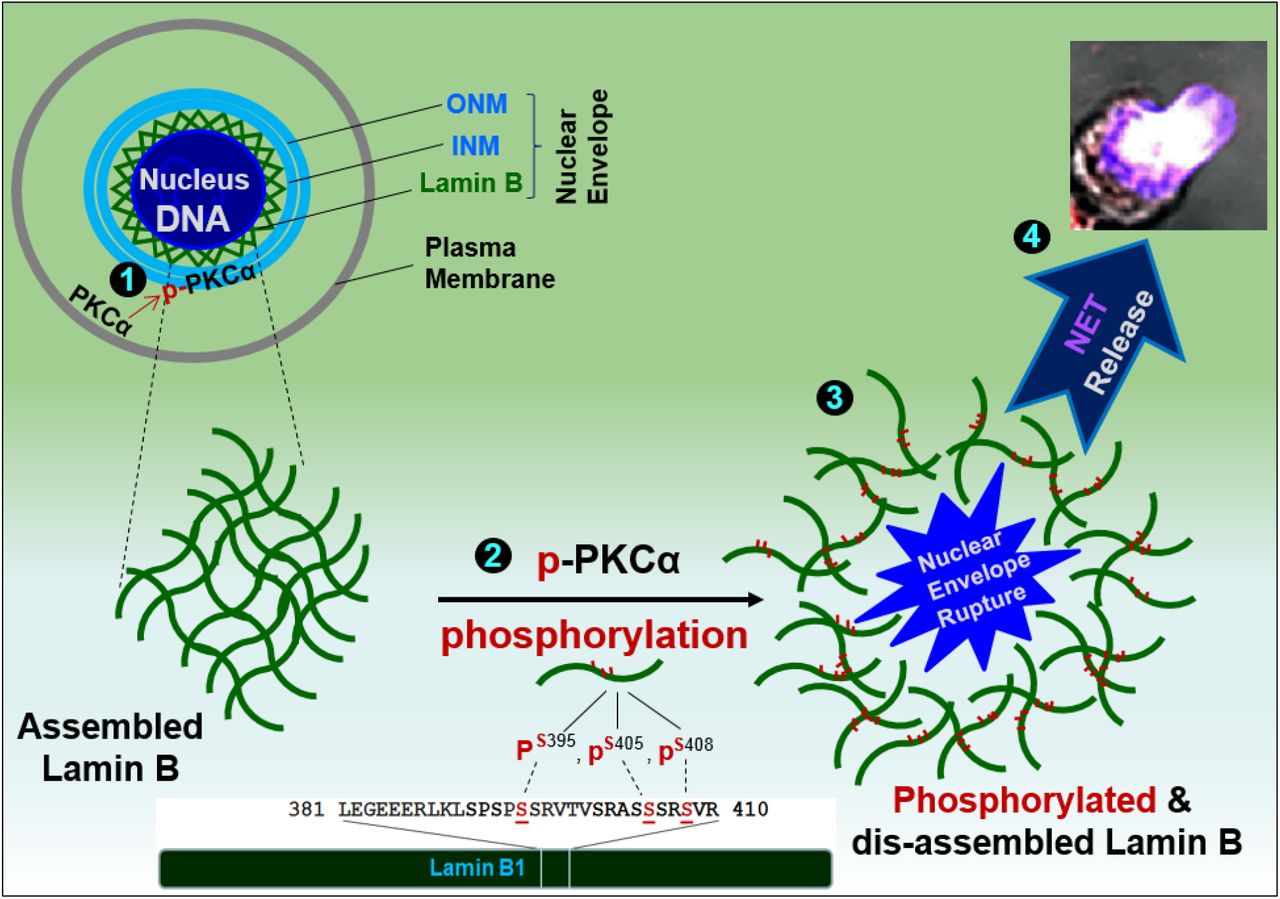
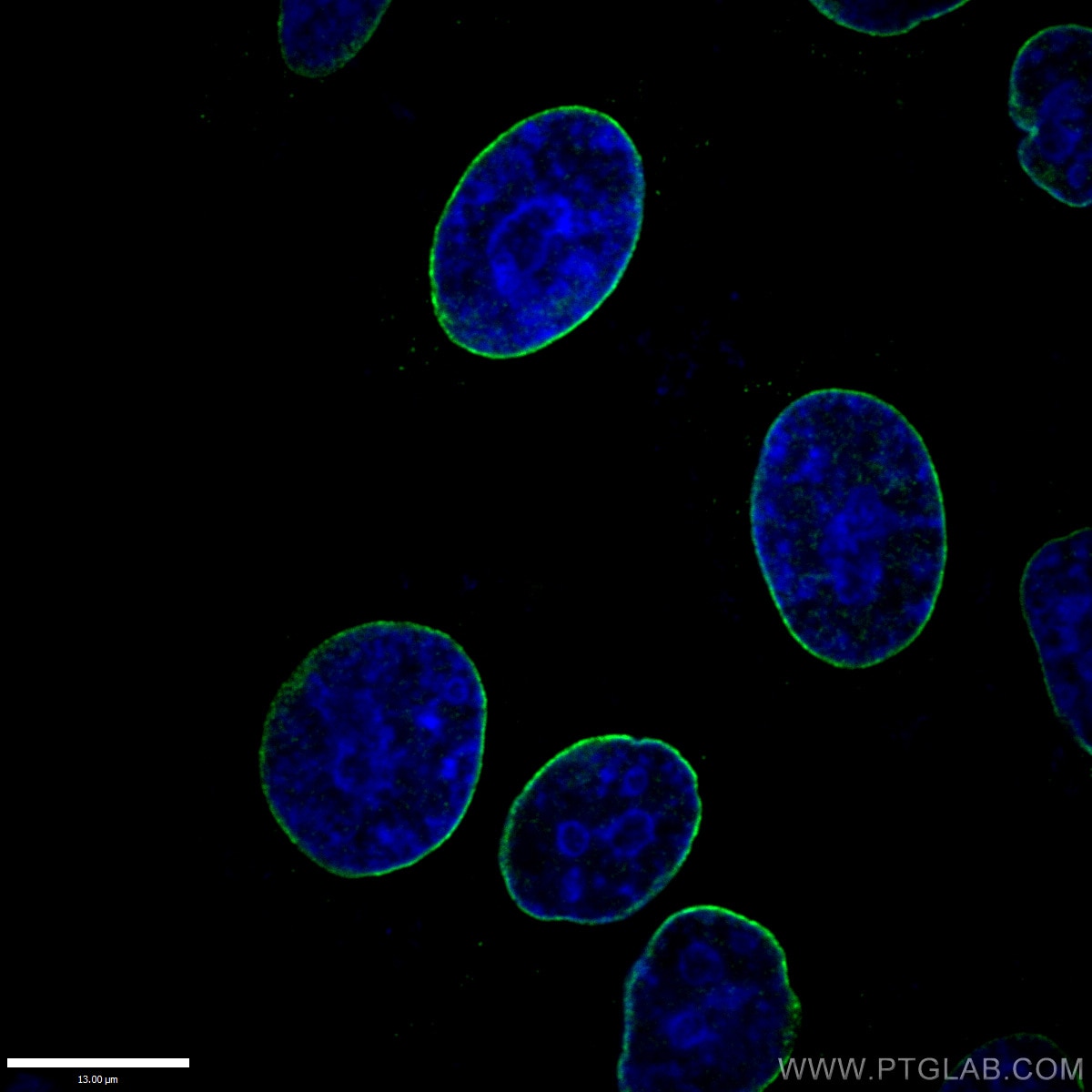
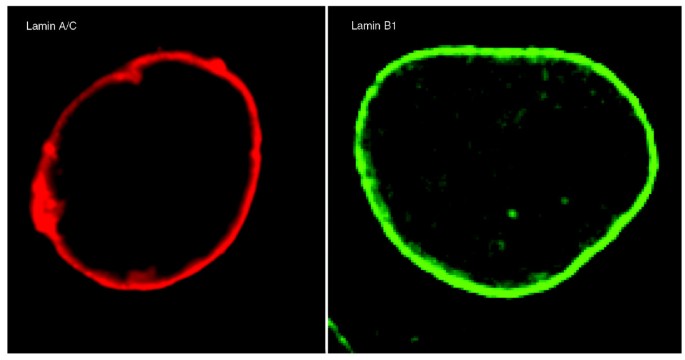
Nuclear lamin B is crucial to the nuclear envelope integrity and extracellular trap release in neutrophils | bioRxiv

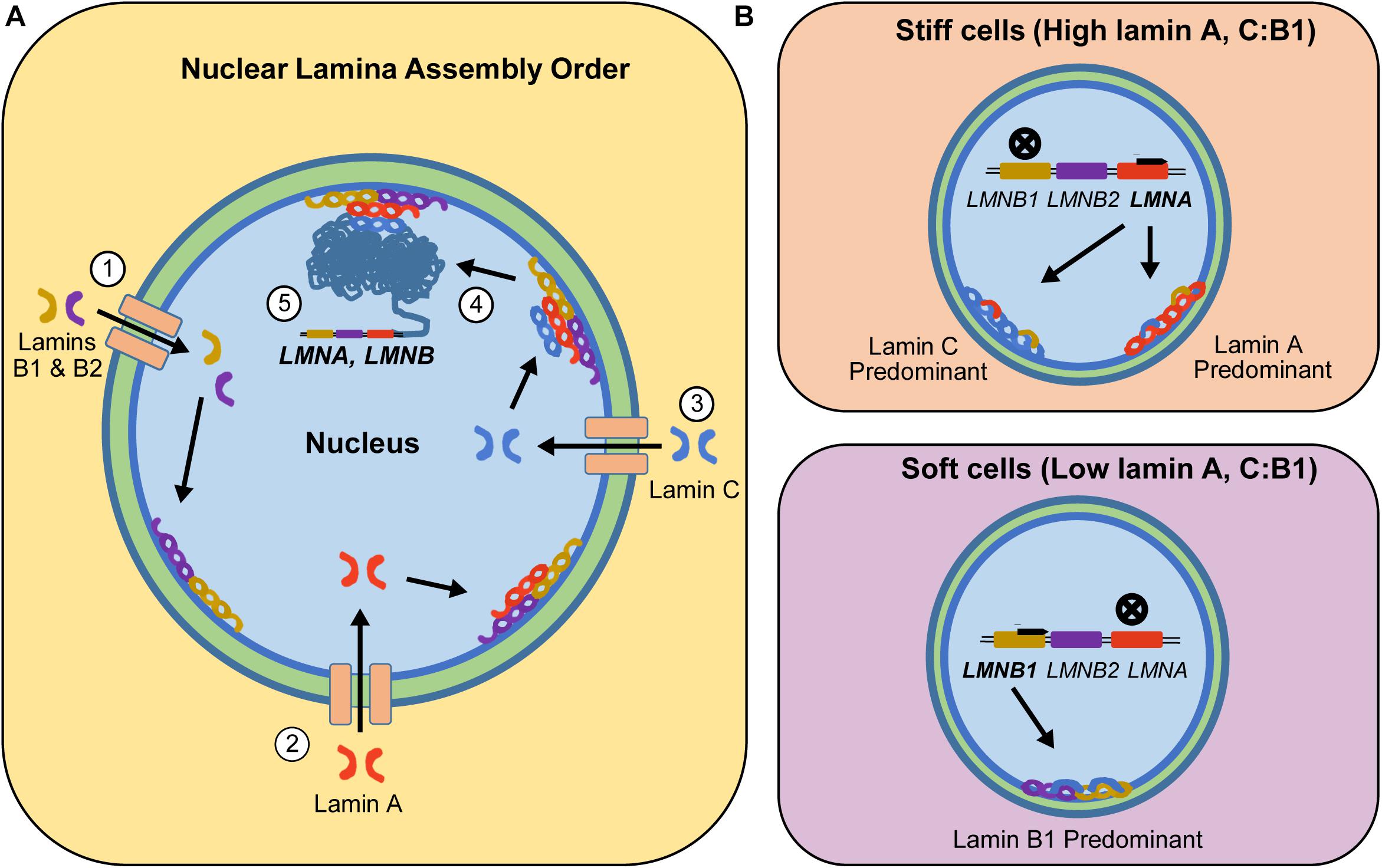
Nuclear lamin isoforms differentially contribute to LINC complex-dependent nucleocytoskeletal coupling and whole-cell mechanics | PNAS